
The isotope shown here is scandium-45, with a nucleus. To determine what "x" is, simply count the number of boxes that you come across before reaching the element you are attempting to determine the electron configuration for. Finally, there are two electrons in the fourth shell (4s orbital). s-orbitals can hold up to a maximum of 2 electrons, p-orbitals can hold up to 6 electrons, d-orbitals can hold up to 10 electrons, and f-orbitals can hold up to 14 electrons). In this case, the "x" in ns x and nd x is the number of electrons in a specific orbital (i.e. However, there is an exception for the d-block and f-block, in which the energy level, "n" for the d block is "n-1" ("n" minus 1) and for the f block is "n-2" (See following periodic table for clarification). Members of a group typically have similar properties and electron configurations in their. 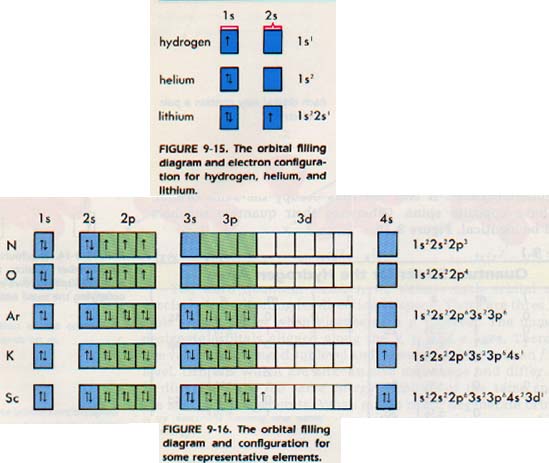
The energy level, "n", can be determined based on the periodic table, simply by looking at the row number in which the element is in. 20 Calcium, Ca, 1s22s22p63s23p64s2 (2.8.8.1), Ar3d 4s 4p 21 Scandium, Sc, 1s22s22p63s23p63d14s Ar3d 4s 4p 22 Titanium, Ti, 1s22s22p63s23p63d24s Ar3d. Element Scandium (Sc), Group 3, Atomic Number 21, d-block, Mass 44.956. In the case of first row transition metals, the electron configuration would simply be 4s x3d x. would be used for the first row transition metals), and the electron configuration would follow a ns xnd x format. The noble gas before the first row of transition metals would be the core written with brackets around the element symbol (i.e. This only applies to the first row transition metals, adjustments will be necessary when writing the electron configuration for the other rows of transition metals. The electron configuration for the first row transition metals consists of 4s and 3d subshells with an argon (noble gas) core. The s, p, d, and f-orbitals are identified on the periodic table below: For this module, we will work only with the first row of transition metals however the other rows of transition metals generally follow the same patterns as the first row. It is a transition metal that belongs to the d-block of the periodic table.One of the key aspects of understanding an element’s behavior and properties is its electron configuration. Scandium is a chemical element with the symbol Sc and atomic number 21. Although the elements can display many different oxidation states, they usually exhibit a common oxidation state depending on what makes that element most stable. Introduction to Scandium Electron Configuration. The electron configuration of transition metals is special in the sense that they can be found in numerous oxidation states. The main focus of this module however will be on the electron configuration of transition metals, which are found in the d-orbitals (d-block). 
The s-orbital primarily denotes group 1 or group 2 elements, the p-orbital denotes group 13, 14, 15, 16, 17, or 18 elements, and the f-orbital denotes the Lanthanides and Actinides group. The s-orbital can hold 2 electrons, and the other three orbitals can hold up to 6, 10, and 14 electrons, respectively. 
All four orbitals can hold different number of electrons. There are four principle orbitals (s, p, d, and f) which are filled according to the energy level and valence electrons of the element. Scandium is the first element to occupy the 3d orbital after filling the 4s orbitals, the first of the "d group" of the transition elements.\) The group is also called the scandium group or scandium family after its lightest member. It is found with yttrium in the silicate mineral Thortveitite. Group 3 is the first group of transition metals in the periodic table.This group is closely related to the rare-earth elements.It contains the four elements scandium (Sc), yttrium (Y), lutetium (Lu), and lawrencium (Lr). Scandium is situated in the transition metal group and has an atomic number of 21. The number of valence electrons available for the Scandium atom is 3. The electron configuration for Scandium ion (Sc 3+) is 1s 2 2s 2 2p 6 3s 2 3p 6. It is usually found in nature with yttrium and lanthanum and the fourteen members of the lanthanide series. The shorthand electron configuration for Scandium is Ar 4s 2 3d 1. It has yet to find much importance in use. Scandium forms colorless compounds similar to those of aluminum.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
